Stryker’s Neuroform Atlas Stent System granted an expanded indication
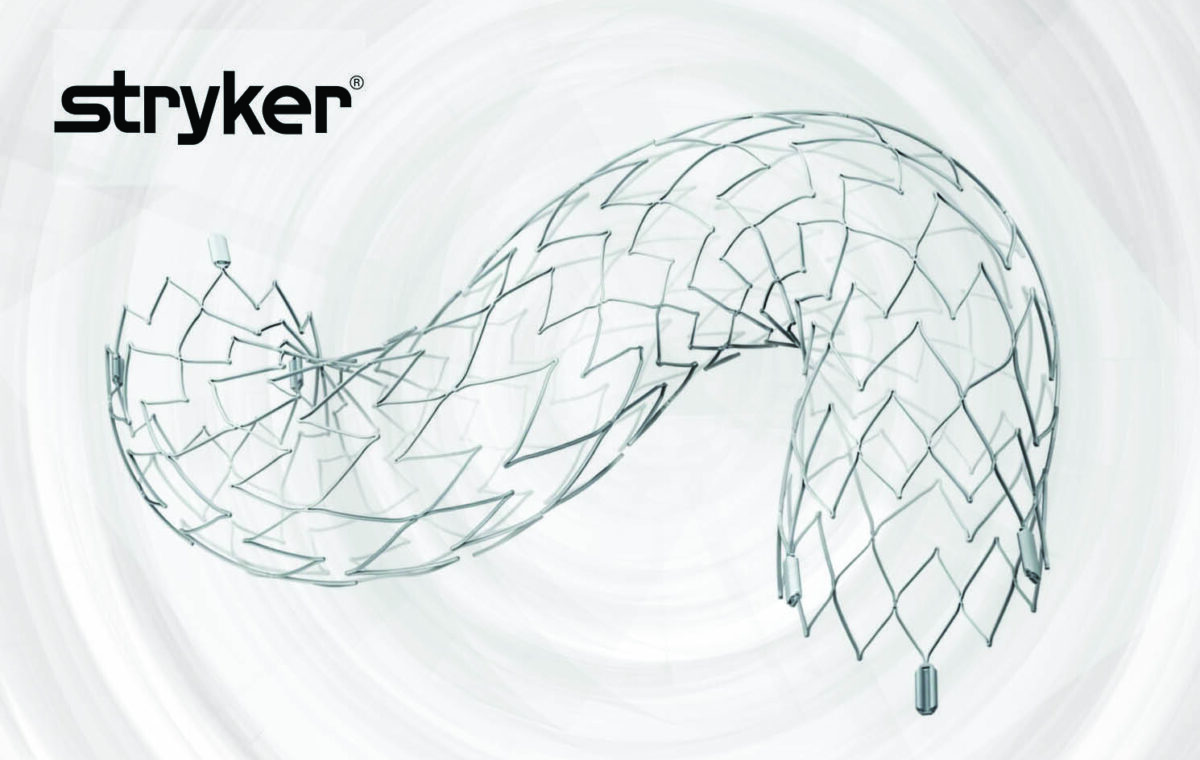
Stryker announced today that it has received U.S. Food and Drug Administration (FDA) approval for an expanded indication of its Neuroform Atlas Stent System, becoming the first and only adjunctive stent approved for use in the posterior (back of the brain) circulation. Aneurysms in the posterior circulation rupture more frequently and are generally more difficult to treat. With the approval of the Neuroform Atlas adjunctive stent for the posterior circulation, long term treatment is more feasible.
Stryker’s Neuroform Atlas® Stent System, which was granted an expanded indication, providing a new option for patients with aneurysms in the back of the brain.
Already approved for use in the anterior circulation, the expanded indication was granted based on robust clinical trial evidence proving the safety and efficacy of the device. The combined patients from both the anterior and posterior cohorts totaled 298 patients, making it the largest study of its kind.
KALAMAZOO, Michigan, USA, Aug. 3, 2020 /PRNewswire


 Brain Aneurysm Foundation
Brain Aneurysm Foundation